Clinical Trials in the UK: Are They Safe, How Do They Work, and Do You Get Paid?
Clinical trials are an importent part of the medical research process, helping doctors and scientists develop new treatments for diseases and conditions. Many people in the UK participate in clinical trials for various reasons, including contributing to medical research, accessing new treatments, or even earning extra money. In this blog, we’ll answer some of the most commonly searched questions like “Are clinical trials safe?”, “Do you get paid for clinical trials?”, and “How do clinical trials work?”.

What Are Clinical Trials?
Clinical trials are research studies involving human participants. They are used to test new treatments, medications, or devices to ensure their safety and effectiveness. Clinical trials are essential in advancing medical knowledge and improving healthcare outcomes. These trials are conducted in various phases to assess a treatment’s safety, dosage, and efficacy.
In the UK, clinical trials are strictly regulated by the Medicines and Healthcare products Regulatory Agency (MHRA)and must meet ethical standards to protect participants. Before enrolling in a trial, volunteers undergo a screening processand informed consent, where all risks and benefits are clearly explained.
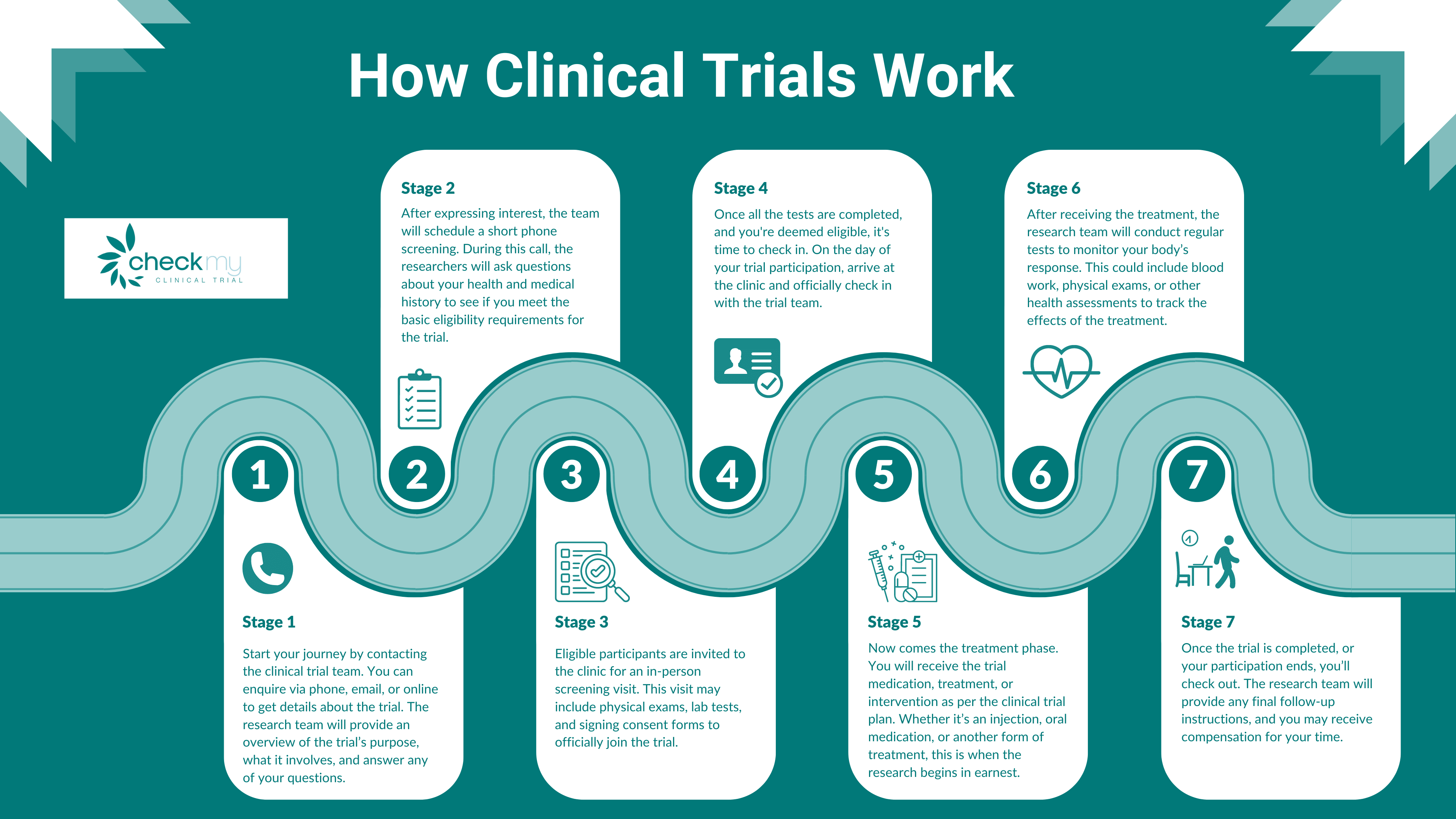
How Do Clinical Trials Work?
Many people wonder, “How do clinical trials work?” Clinical trials are research studies involving human participants to evaluate the safety and effectiveness of new drugs, treatments, or devices. They are conducted in four phases:
Do You Get Paid for Clinical Trials?
Yes, many clinical trials in the UK offer compensation, especially for early-phase trials like Phase I studies. Compensation can vary depending on the type and length of the trial, as well as the level of risk involved. For example, some drug trials pay participants for their time, travel, and inconvenience, with more complex or riskier trials offering higher pay.
Common search queries related to compensation include:
- “How much do you get paid for clinical trials?”
- “Do you get paid for Synexus trials?”
- “How much do you get paid for being a test subject?”
Typically, participants can earn anywhere from a few hundred to several thousand pounds, depending on the complexity of the trial. For more information on available trials and potential compensation, websites like Be Part of Research offer detailed listings of clinical trials in the UK.
What Are the 4 Phases of Clinical Trials?
Understanding the four phases of clinical trials is crucial for those considering participation:
Phase IV: Conducted after a drug or treatment has been approved. This phase looks at long-term safety and effectiveness across a broader population.
Phase I: Focuses on safety, determining the right dosage, and identifying side effects. These trials are often done with a small group of healthy volunteers.
Phase II: Examines the drug’s efficacy, continuing to monitor safety. This phase involves more participants who have the condition the drug is meant to treat.
Phase III: Involves thousands of patients to confirm effectiveness, monitor side effects, and compare the new treatment to existing ones.

Are Clinical Trials Safe?
One of the most frequently asked questions is: “Are clinical trials safe?” While clinical trials are designed with safety in mind, they do carry some risks, especially in early phases like Phase I, where the treatment or drug is being tested on humans for the first time.
However, clinical trials in the UK are strictly regulated by the MHRA and follow stringent ethical guidelines. Participants are required to go through an informed consent process, where they are made fully aware of any potential risks or side effects. This ensures participants can make an informed decision about whether or not to participate.
In addition to informed consent, clinical trials are continuously monitored by independent review boards (IRBs) and ethics committees that ensure the trial follows all safety protocols.
It’s also important to note that later phases of clinical trials (like Phase III and Phase IV) involve far less risk, as the treatment has already been tested on larger groups of people.
What Are the Risks of Clinical Trials?
“Are clinical trials safe?” is a common question for a reason. While most clinical trials are designed to be as safe as possible, there are still risks involved, particularly in early-phase trials. These risks can include:
- Side Effects: Clinical trials often test new drugs or treatments that have not yet been fully evaluated, meaning participants could experience side effects, ranging from mild (like headaches or nausea) to severe (such as allergic reactions).
- Ineffectiveness: There’s also the chance that the treatment being tested will not work as intended or may be less effective than current treatments.
- Placebo Effect: In some trials, participants are given a placebo (an inactive substance) instead of the actual treatment. This means you may not receive the treatment being tested, which could be frustrating if you’re hoping to access new therapies.
However, strict monitoring and safety protocols ensure that participants’ well-being is prioritized throughout the trial process.

How Do I Join a Clinical Trial in the UK?
If you’re wondering, “How do I join a clinical trial?”, the process is straightforward:
Participation: Once you’re approved, you’ll begin the trial. Depending on the phase and nature of the study, this could involve anything from taking medication to regular medical checkups or even overnight stays in a medical facility.
Find a Trial: There are several online platforms that list ongoing trials, such as ClinicalTrials.gov and NIHR BePart of Research. You can search for trials based on your location, medical condition, or interest in specific treatments.
Eligibility: Each trial has specific inclusion and exclusion criteria, such as age, gender, medical history, and current health status. Make sure to review these criteria to see if you’re eligible for the trial.
Screening Process: If you’re eligible, you’ll undergo a screening process, which may include a medical examination, blood tests, or other assessments to ensure you’re a good fit for the trial.
Informed Consent: Before participating, you’ll need to sign an informed consent form, which outlines all the details of the trial, including potential risks and benefits.

Leave a comment